1. Increasing state control over digital spaces raises ethical concerns regarding autonomy and surveillance. Discuss the ethical implications of prolonged data retention and content regulation.
| Syllabus: Ethics – General Studies – : IV Probity in Governance: Concept of public service; Philosophical basis of governance and probity; Information sharing and transparency in government, Right to Information, Codes of Ethics, Codes of Conduct, Citizen’s Charters, Work culture, Quality of service delivery, Utilization of public funds, challenges of corruption. |
IN NEWS: A tightening of the fist in India’s digital public square
The expansion of state control over digital spaces creates a fundamental tension between national security and individual liberty. As governments implement more stringent data retention mandates and content regulation frameworks, the ethical landscape shifts from one of digital “openness” to one of “managed environments.”
1. Ethical Implications of Prolonged Data Retention
Prolonged data retention involves the mandatory storage of metadata and communication records for extended periods (often years). This practice raises several ethical dilemmas:
- The Panopticon Effect and Self-Censorship: When individuals are aware that their digital footprint—searches, locations, and messages—is permanently recorded, they may alter their behavior. This “chilling effect” erodes autonomy; the mere possibility of surveillance can lead to self-regulation, where citizens avoid controversial topics or dissent to stay within perceived “safe” boundaries.
- Purpose Creep: Data collected for a specific, narrow purpose (e.g., stopping a specific crime) is often repurposed for broader social sorting or political monitoring. Ethically, this violates the principle of Informed Consent, as the scope of data use expands without the user’s active agreement.
- Security vs. Privacy Paradox: While governments argue that long-term data logs are essential for forensic investigations, the existence of massive centralized databases creates “honeypots” for hackers. The ethical risk here is that in the pursuit of state security, the individual’s data security is actually diminished.
2. Ethical Implications of Content Regulation
Content regulation involves state-mandated filtering, age-gating, or the removal of “objectionable” material. The ethical concerns center on agency and transparency:
- Paternalism vs. Autonomy: Strict content regulation, such as mandatory age-verification or “decency” filters, often acts as a form of state paternalism. While intended to protect the vulnerable, it can restrict the right of adults to access information and engage in creative expression.
- The “Vagueness” Problem: Regulations often use subjective terms like “misleading,” “obscene,” or “anti-national.” Without precise definitions, these laws grant executive bodies significant discretion. This lack of predictability is ethically problematic because it allows for arbitrary enforcement, often targeting political dissent or minority views under the guise of “public order.”
- Algorithmic Bias and Accountability: States increasingly pressure platforms to use AI for automated content removal. These algorithms often lack the nuance to distinguish between satire, news, and actual harm. The ethical failure here lies in the lack of due process; when content is “disappeared” by a machine, the user often has no transparent path for appeal or redress.
Ethical frameworks suggest that for state intervention to be legitimate, it must meet a three-part test: Legality (clear laws), Necessity (the least intrusive means), and Proportionality (the benefit to society must outweigh the harm to individual privacy). Without these safeguards, the digital space risks transitioning from a tool of empowerment to one of systemic control.
2. Cutting-edge therapies like CAR-T are often expensive and complex. Analyse the challenges in making such advanced treatments accessible in developing countries like India.
| Syllabus: Science and technology – General Studies – : III Science and Technology- developments and their applications and effects in everyday life. |
IN NEWS: CAR-T cell therapy senses ‘faint’ targets to clear solid tumours
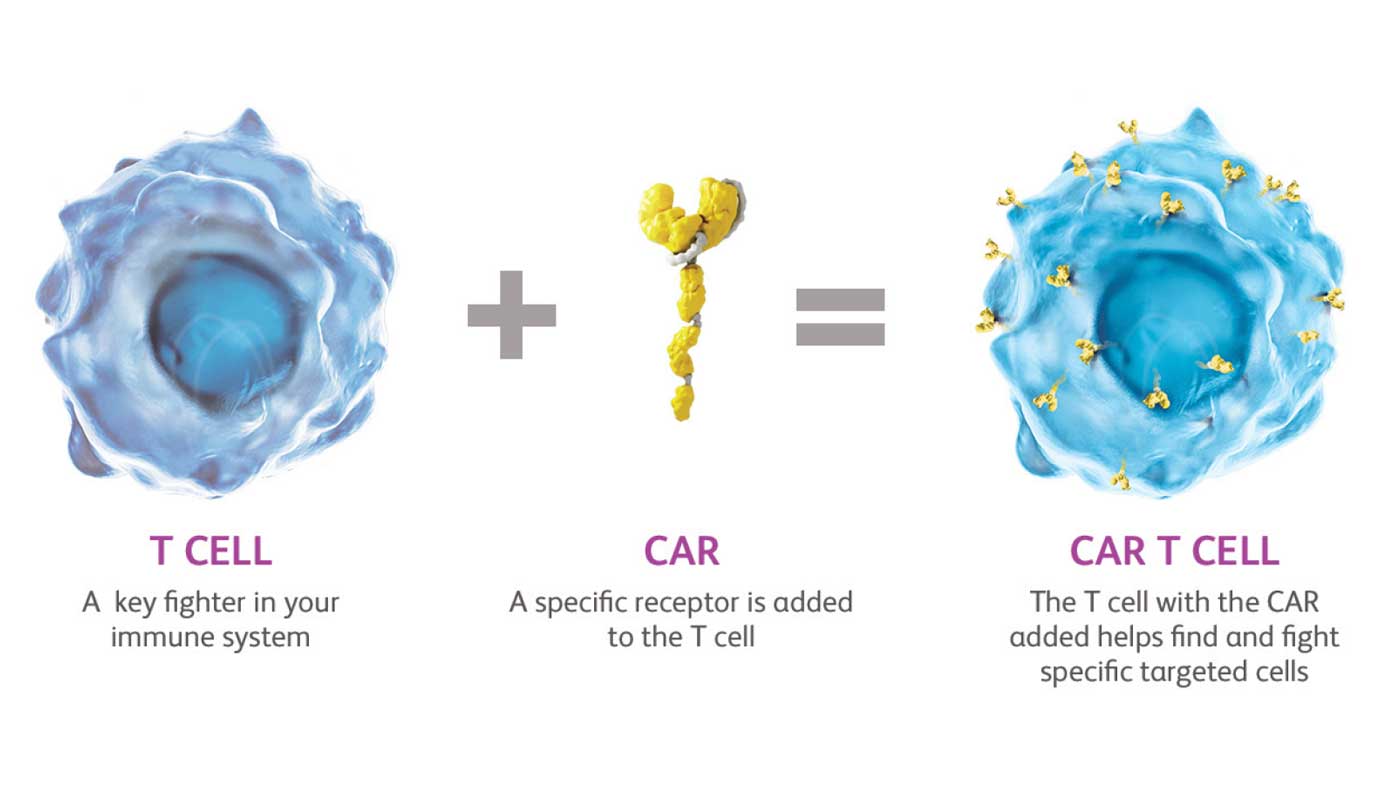
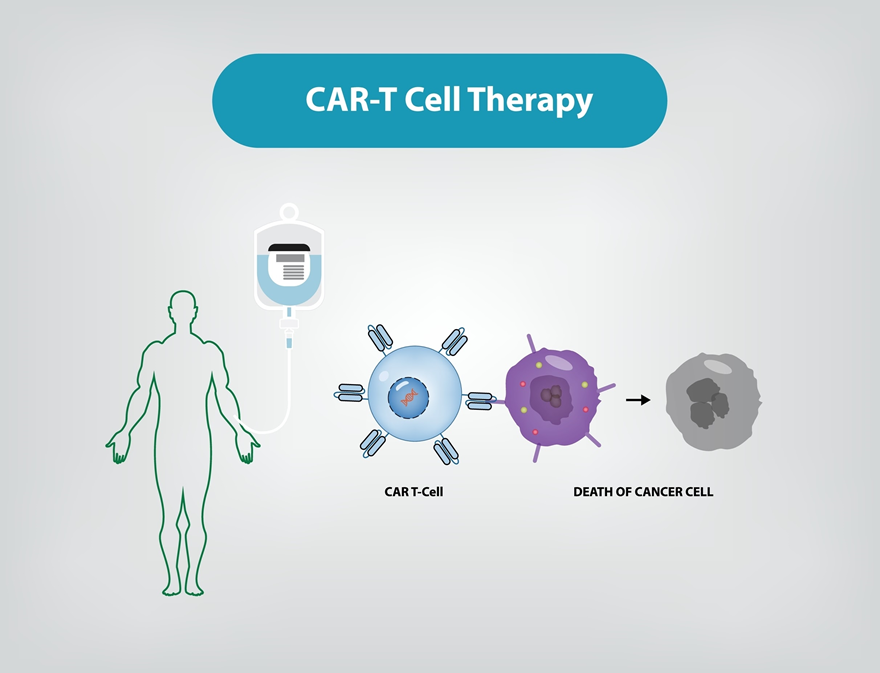
The challenge of making cutting-edge therapies like Chimeric Antigen Receptor T-cell (CAR-T) accessible in developing nations like India is a classic example of the “innovation-access gap.” While India has made historic strides—such as the 2024 launch of its indigenous CAR-T therapy, NexCAR19—deep-seated structural hurdles remain.
1. The Cost Paradox
Although India has managed to reduce the cost of CAR-T therapy significantly, it remains out of reach for the majority.
- Global vs. Local Pricing: In the US, CAR-T costs upwards of $400,000 (~₹3.3 Crore). India’s indigenous version, NexCAR19, is priced at roughly ₹30–40 Lakh.
- Affordability Gap: Despite the 90% reduction, the cost is still nearly 20 times the per capita GDP of India. With limited private insurance coverage and virtually no public reimbursement for such high-end therapies, patients often face catastrophic out-of-pocket expenses.
2. Infrastructure and Logistical Complexity
CAR-T is not a “pill” but a “living drug” that requires a sophisticated, sterile supply chain.
- Personalized Manufacturing: Unlike mass-produced vaccines, CAR-T requires extracting a patient’s own T-cells, genetically modifying them in a lab, and re-infusing them. This necessitates GMP-certified (Good Manufacturing Practice) facilities that are currently concentrated only in Tier-1 cities like Mumbai, Delhi, and Bengaluru.
- The Cold Chain Challenge: Modified cells must be transported and stored at cryogenic temperatures (often -80°C or lower). Maintaining this “vein-to-vein” integrity across India’s vast and varied climate is a major logistical hurdle.
3. Clinical and Tertiary Care Requirements
Administering CAR-T is high-risk and requires specialized medical ecosystems.
- Toxicity Management: Patients can suffer from Cytokine Release Syndrome (CRS) or neurotoxicity, which requires immediate access to advanced ICUs and expensive “rescue” drugs like Tocilizumab.
- Skilled Human Capital: The therapy requires a multidisciplinary team of hematologists, intensivists, and specialized lab technicians—a workforce that is currently stretched thin in the public sector.
The “India Model” for the Future
To bridge these gaps, India is pursuing a strategy of “frugal innovation”:
- Decentralized Manufacturing: Moving production closer to hospitals to reduce transport costs.
- Humanized Constructs: NexCAR19 uses a more “human-like” antibody to reduce severe side effects, thereby lowering the need for expensive ICU stays.
- Public-Private Partnerships: Collaborative efforts between institutions like IIT Bombay, Tata Memorial Centre, and industry partners (like ImmunoACT) to scale production.
Making CAR-T accessible in developing countries requires moving beyond “price cuts” toward ecosystem building. Success depends on whether the state can integrate these therapies into public health schemes (like Ayushman Bharat) and whether localized manufacturing can further bring costs down to the target range of ₹10–15 Lakh.
| PYQ REFERENCE (UPSC 2021) Q. “What are the research and developmental achievements in applied biotechnology? How will these achievements help to uplift the poorer sections of the society?” |